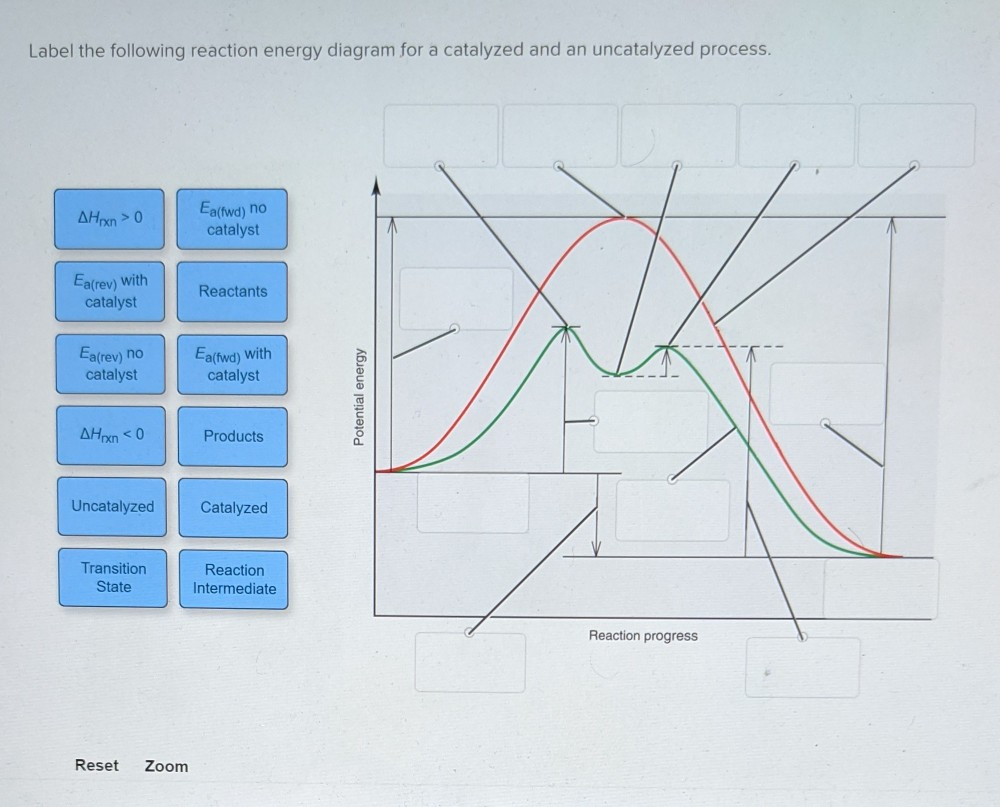
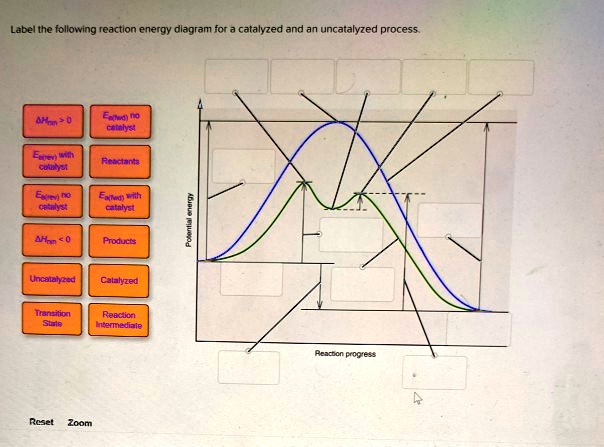
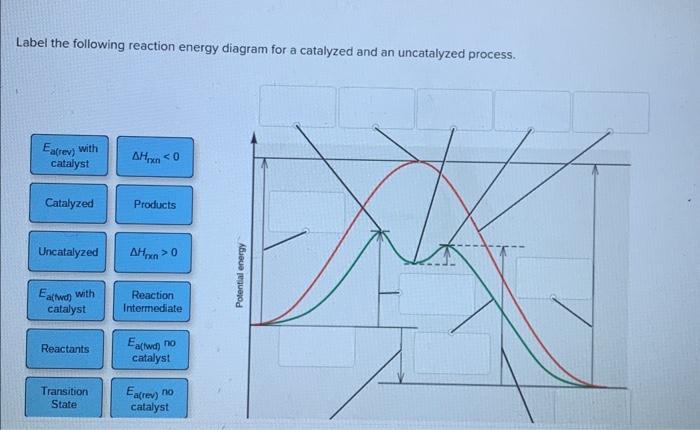
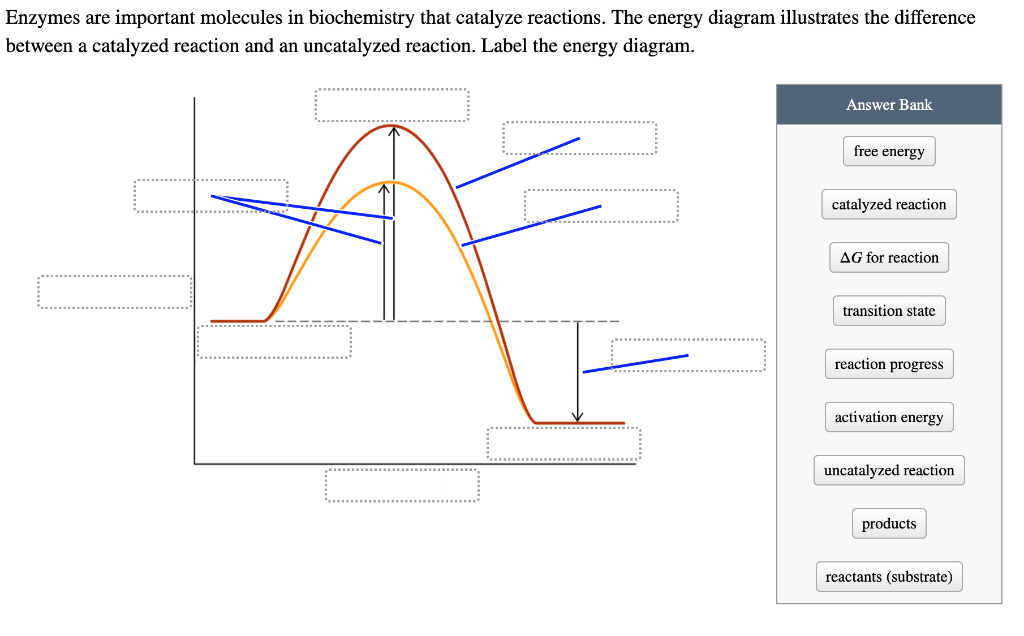
44 label the following reaction energy diagram for a catalyzed and an uncatalyzed process
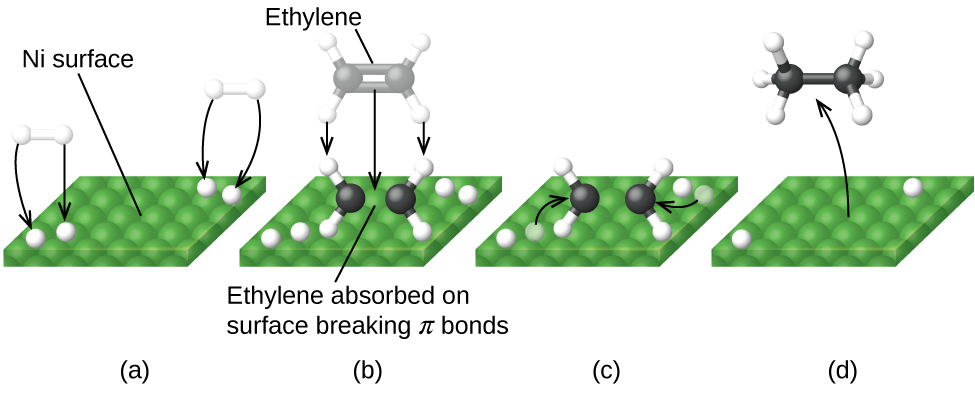
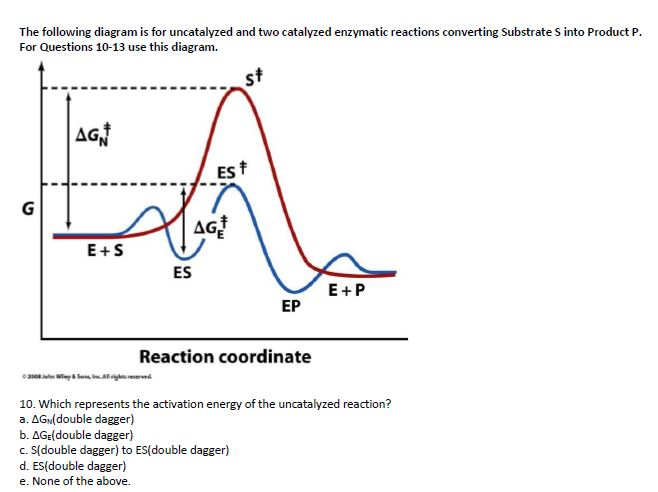
label the following reaction energy diagram for a catalyzed and an ... Take a look at this or that energy diagram. In the case of the uncatalyzed reaction, the more energy you have, the more energy you can save for the reaction. The catalyst is the element of life, zinc. Zinc is the fourth element in the periodic table. It is the element that enables life to occur. BioChemistry Ch 8 Flashcards The lock-and-key model explains the steps involved in an enzyme-catalyzed reaction. Label the following diagram that illustrates the lock-and-key model of enzyme activity. Classify each of the characteristics as lock-and-key model or induced-fit model or both. The enzyme urease catalyzes the breakdown of urea in the body.
12.7: Catalysis - Chemistry LibreTexts Figure 12.7.1: This graph compares the reaction coordinates for catalyzed and uncatalyzed alkene hydrogenation. A graph is shown with the label, "Reaction coordinate," on the x-axis and the label,"Energy," on the y-axis.
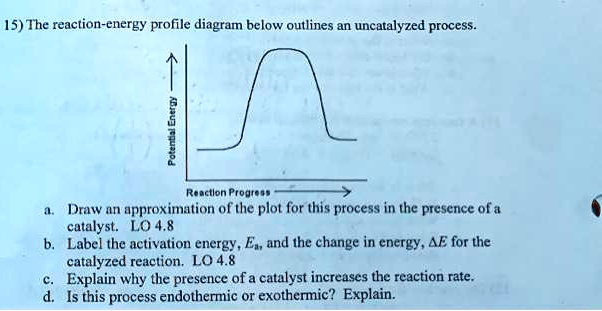
Label the following reaction energy diagram for a catalyzed and an uncatalyzed process
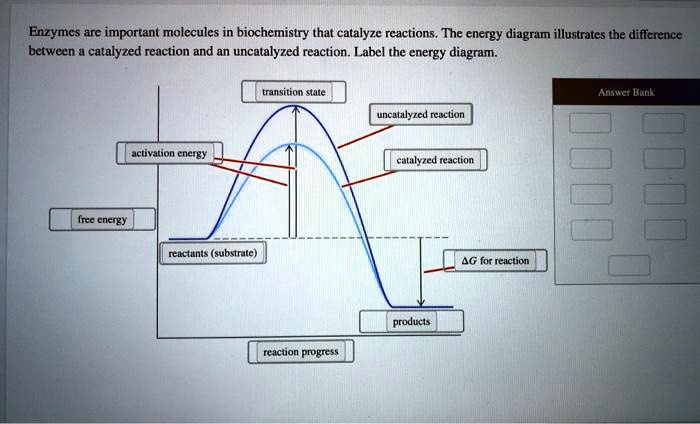
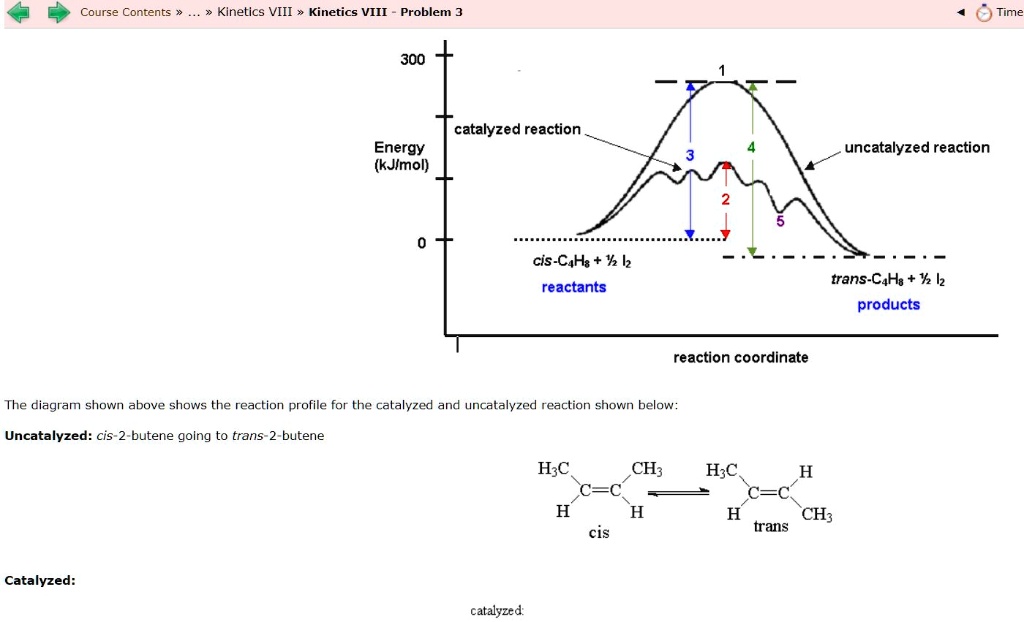
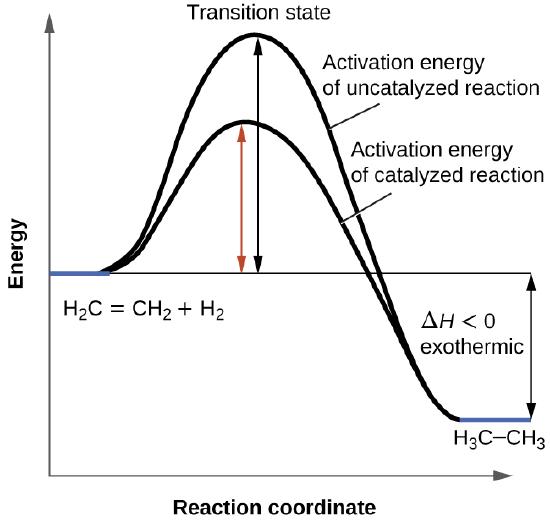
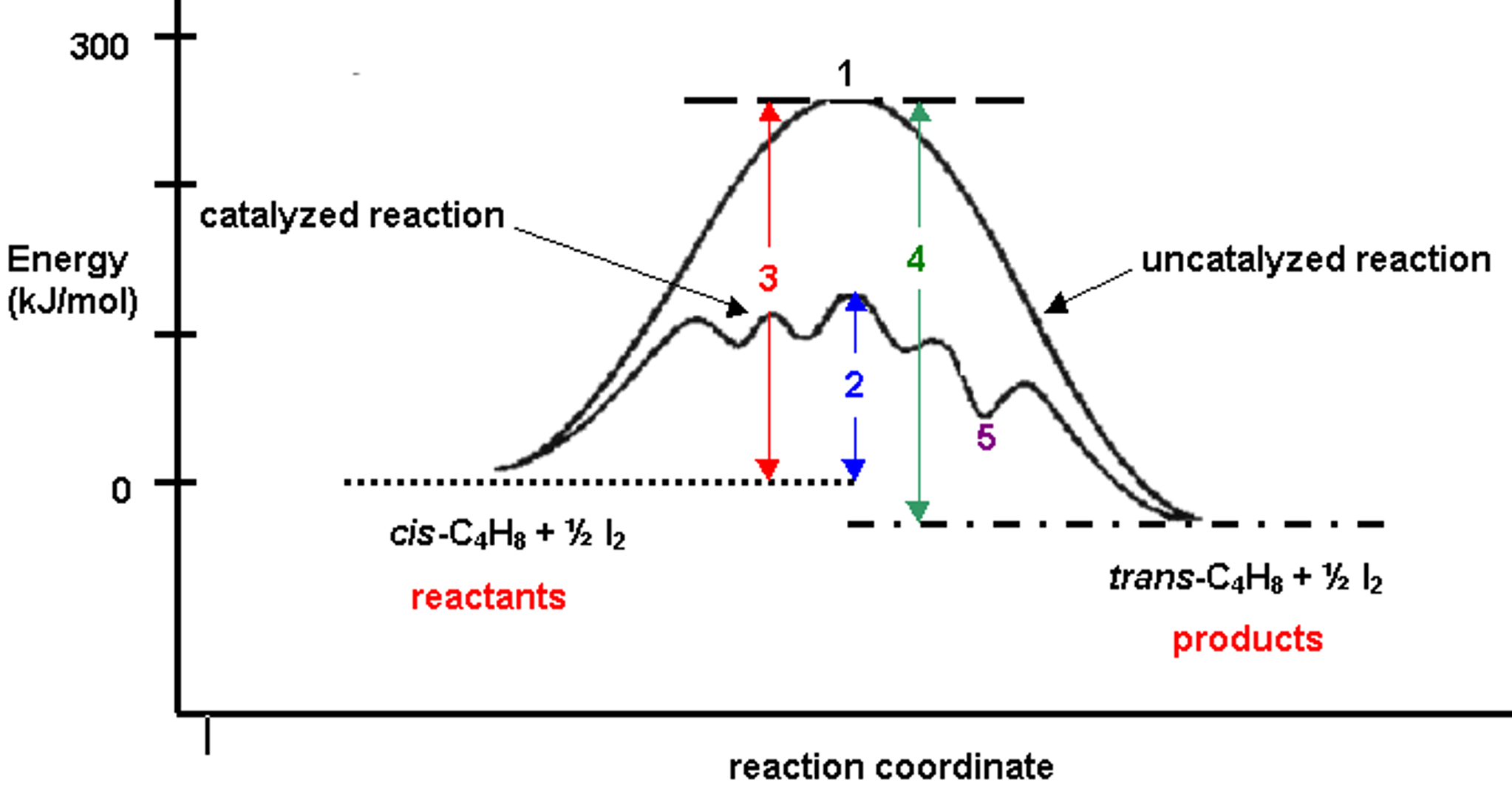
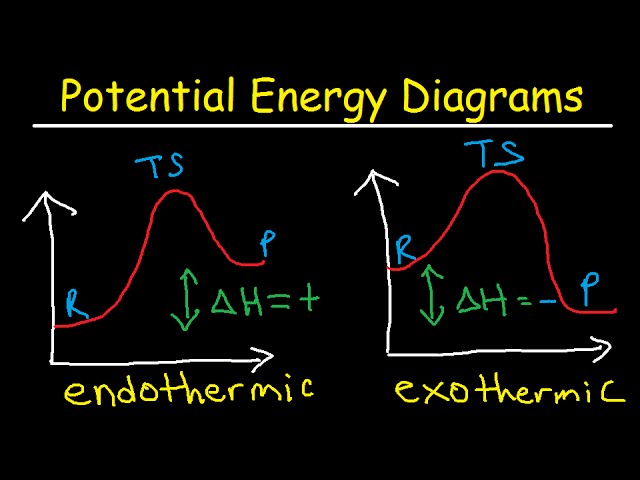
12.7 Catalysis - Chemistry 2e | OpenStax For the first step, Ea = 80 kJ for (a) and 70 kJ for (b), so diagram (b) depicts the catalyzed reaction. Activation energies for the second steps of both mechanisms are the same, 20 kJ. Homogeneous Catalysts A homogeneous catalyst is present in the same phase as the reactants. Types of catalysts (article) | Kinetics | Khan Academy Notice that the energies of the reactants and products are the same for the catalyzed and uncatalyzed reaction. Therefore, the overall energy released during the reaction, Δ H rxn \Delta \text H_{\text{rxn}} Δ H rxn delta, start text, H, end text, start subscript, start text, r, x, n, end text, end subscript, does not change when you add the enzyme. . This emphasizes a very important point ... Potential Energy Diagram of Catalyzed and Uncatalyzed Reactions 3.9K views 2 years ago #narcademy Analyzing the potential energy diagram of a regular/uncatalyzed and a catalyzed (adding a catalyst) reaction. Remember that the 🔼H of reaction remains...
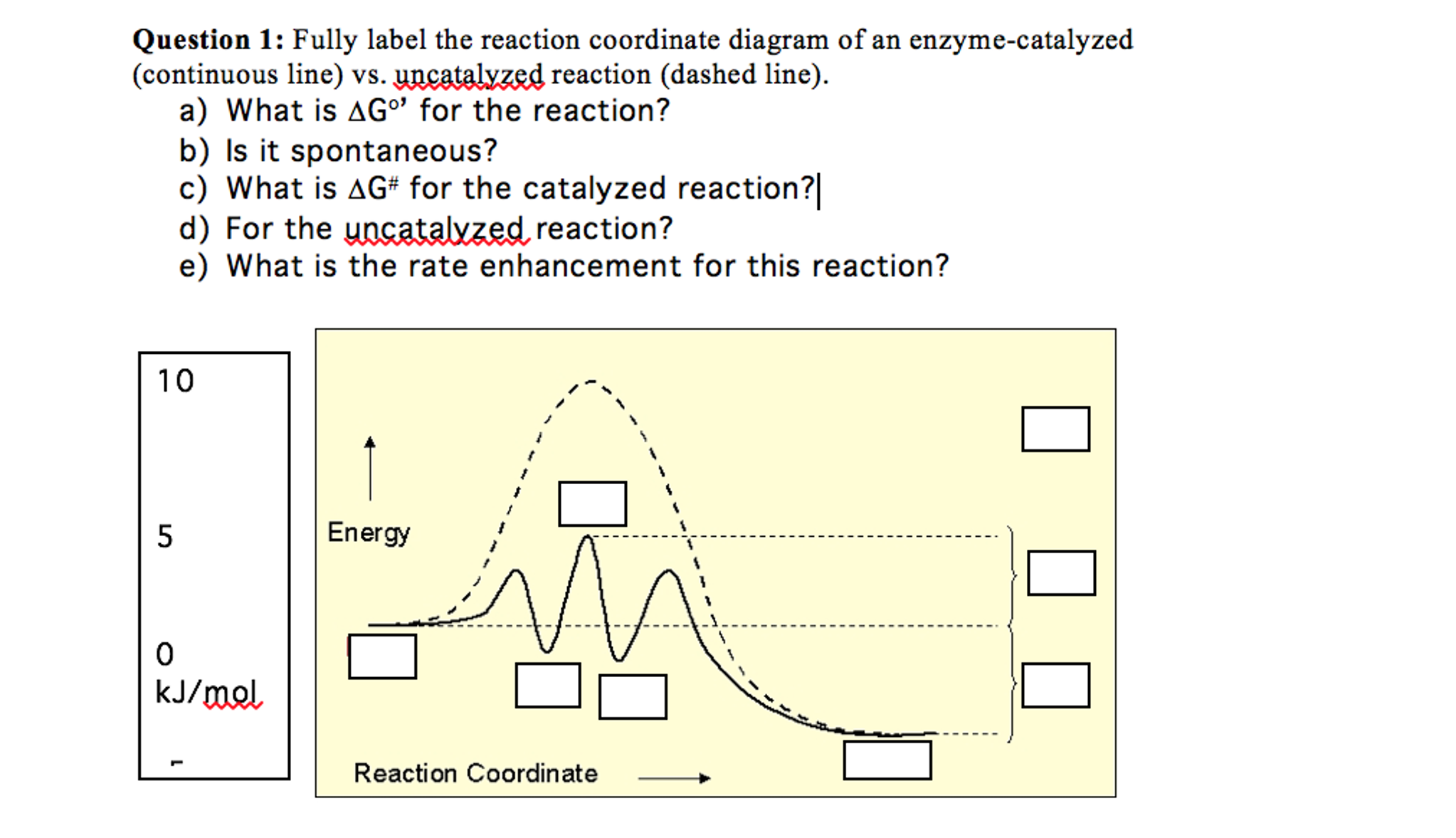
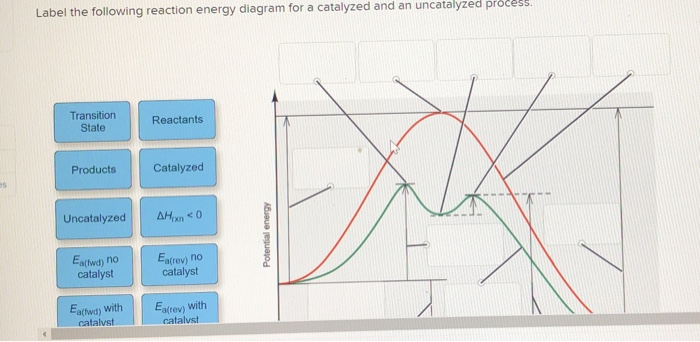
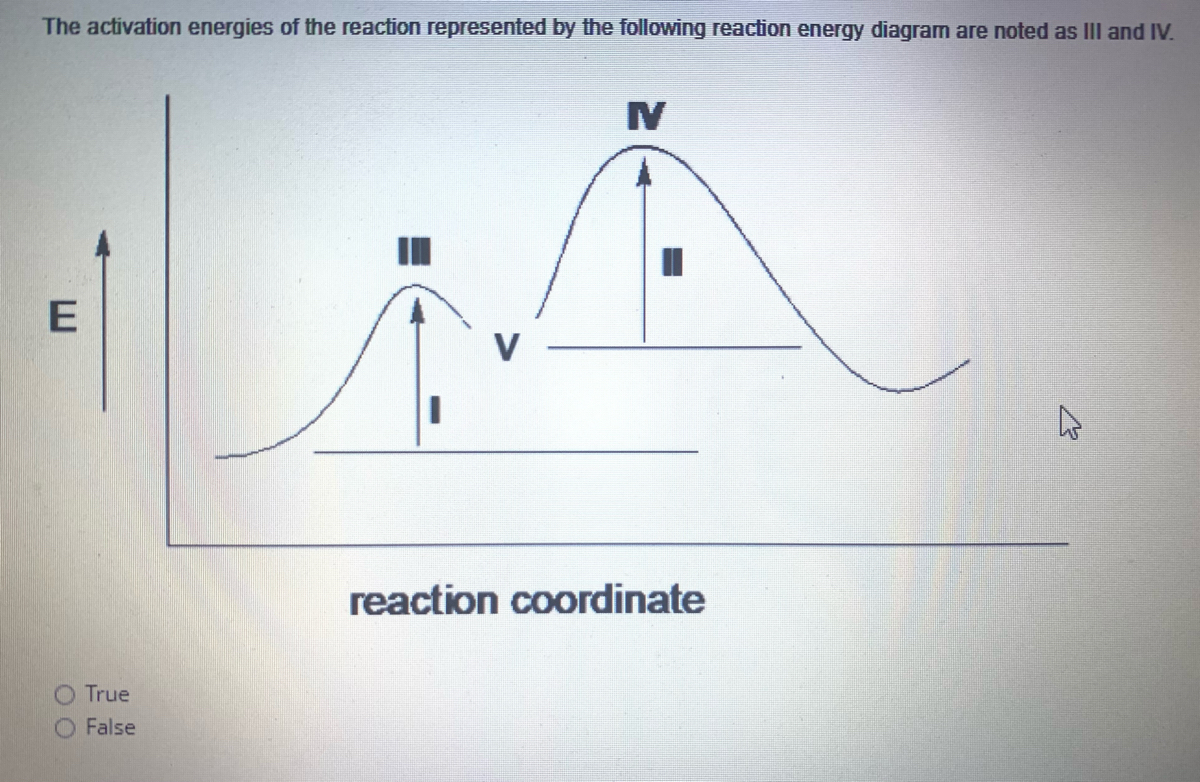
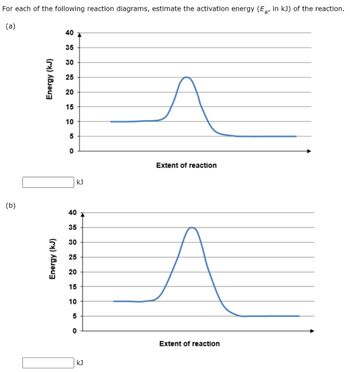
Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. Catalysis | Chemistry for Majors Example 1: Using Reaction Diagrams to Compare Catalyzed Reactions. The two reaction diagrams here represent the same reaction: one without a catalyst and one with a catalyst. Estimate the activation energy for each process, and identify which one involves a catalyst. Solved Label the following reaction energy diagram for a | Chegg.com Transition State Transition State Transition intermediate Reaction Intermediate Transition State Transition Uncatalyzed Transition State Reactants Products Catalyzed Ea (fwd) no catalyst Uncatalyzed AHxn < 0 Potential energy Ea (rev) no catalyst Ea (fwd) no catalyst Ea (rev) Show transcribed image text Expert Answer 100% (79 ratings) Chem Chapter 10 Homework 1 Flashcards The reaction energy diagrams for four reactions are given. Arrange the reactions according to their relative rates of reaction. Check all that apply The activation energy is higher in an uncatalyzed reaction than in an enzyme-catalyzed reaction. An enzyme-substrate complex (ES) is formed in an enzyme-catalyzed reaction. 12.7 Catalysis | Chemistry One such reaction is catalytic hydrogenation, the process by which hydrogen is added across an alkene C=C bond to afford the saturated alkane product. A comparison of the reaction coordinate diagrams (also known as energy diagrams) for catalyzed and uncatalyzed alkene hydrogenation is shown in Figure 1. Figure 1.
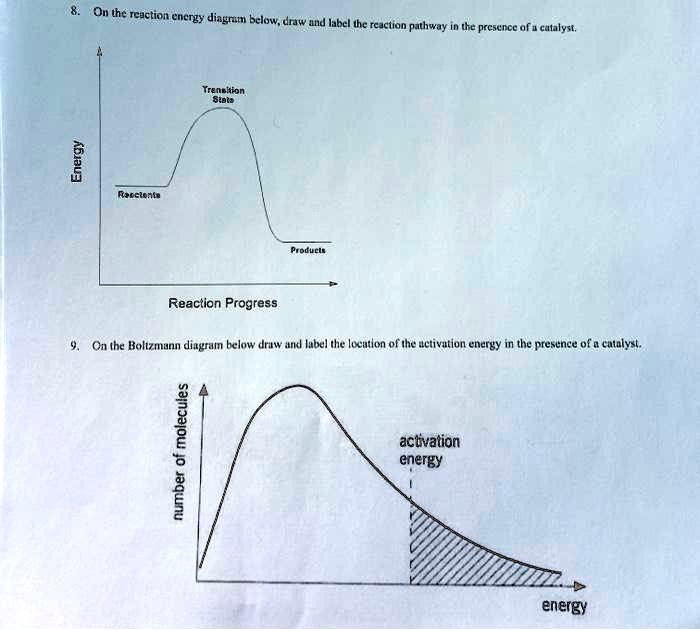
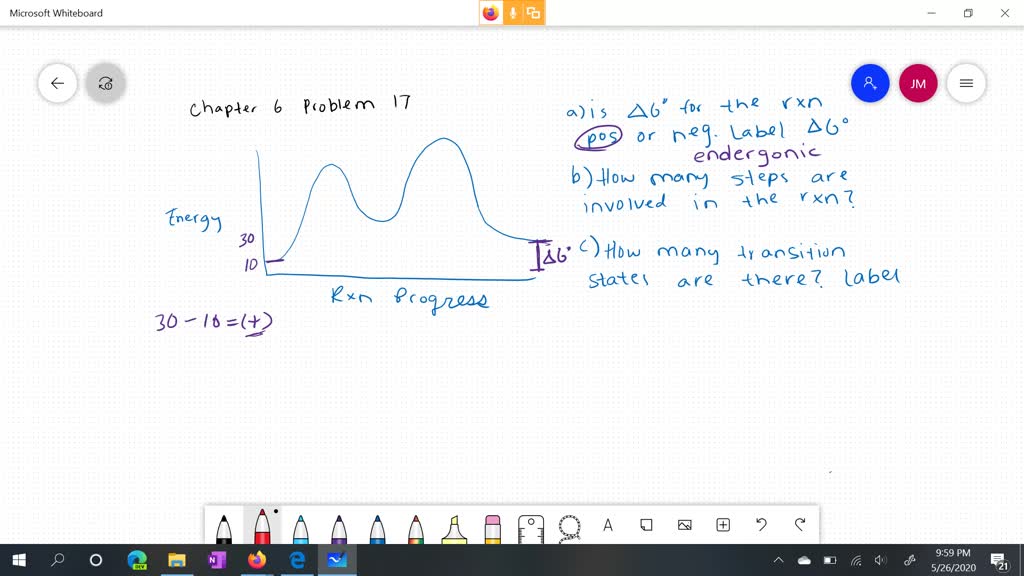
14.7: Catalysis - Chemistry LibreTexts This graph compares potential energy diagrams for a single-step reaction in the presence and absence of a catalyst. The only effect of the catalyst is to lower the activation energy of the reaction. The catalyst does not affect the energy of the reactants or products (and thus does not affect ΔE). (CC BY-NC-SA; anonymous) Drawing the Reaction Energy Diagram of a Catalyzed Reaction Steps for Drawing the Reaction Energy Diagram of a Catalyzed Reaction. Step 1: Determine the energies of the reactants and products and the reaction enthalpy. Step 2: Determine the activation ... Catalysis - Chemistry Catalysts function by providing an alternate reaction mechanism that has a lower activation energy than would be found in the absence of the catalyst. In some cases, the catalyzed mechanism may include additional steps, as depicted in the reaction diagrams shown in [link]. This lower activation energy results in an increase in rate as described ... Solved Label the following reaction energy diagram for a | Chegg.com Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. Uncatalyzed Catalyzed Eare) with catalyst Ea {fwd) with catalyst 1 Products AHex > 0 -- - Potential energy Transition State AHp <0 Reaction Intermediate Reactants Ea (rev no catalyst Ealfwd) no catalyst Reaction progress Previous question Next question
Potential Energy Diagram of Catalyzed and Uncatalyzed Reactions 3.9K views 2 years ago #narcademy Analyzing the potential energy diagram of a regular/uncatalyzed and a catalyzed (adding a catalyst) reaction. Remember that the 🔼H of reaction remains... Types of catalysts (article) | Kinetics | Khan Academy Notice that the energies of the reactants and products are the same for the catalyzed and uncatalyzed reaction. Therefore, the overall energy released during the reaction, Δ H rxn \Delta \text H_{\text{rxn}} Δ H rxn delta, start text, H, end text, start subscript, start text, r, x, n, end text, end subscript, does not change when you add the enzyme. . This emphasizes a very important point ... 12.7 Catalysis - Chemistry 2e | OpenStax For the first step, Ea = 80 kJ for (a) and 70 kJ for (b), so diagram (b) depicts the catalyzed reaction. Activation energies for the second steps of both mechanisms are the same, 20 kJ. Homogeneous Catalysts A homogeneous catalyst is present in the same phase as the reactants.




















Post a Comment for "44 label the following reaction energy diagram for a catalyzed and an uncatalyzed process"